CLIA Lab Service for COVID-19 Diagnostics
Better Accuracy & Faster Turnaround Time for your Back to Work & Back to School Testing
DiaCarta CLIA Lab has developed a COVID-19 Diagnosis Test utilizing the FDA EUA approved QuantiVirus™ SARS-CoV-2 Test, a highly sensitive RT-PCR assay that detects the presence of the virus genes in the samples and therefore increases sensitivity and testing accuracy. This cost-effective SARS-CoV-2 test reduces the workload from major commercial labs and hospitals and delivers fast results within 48 hours of receiving the samples.
Service Highlights
- Electronic or manual accessioning of patient data
- Automated RNA extraction
- High throughput PCR cyclers
- Automated test results generation
- Automated result transmission to customers
- Turnaround time within 48 hours on receipt of samples
- Lab capability is 1,000 samples per day and increasing
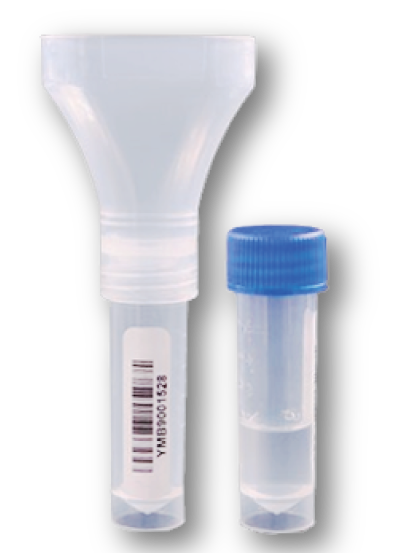
Sample Collection Kits Available
- Nasal sample collection kit. Learn More>
- Saliva sample collection kit. Learn more>
FDA EUA Approved Test
QuantiVirus™ SARS-CoV-2 Test Kit. Learn More>
COVID-19 Hotline
Email: covid19support@diacarta.com
Phone #: +1 510-878-6662, option 4 (tech support)
GET A QUOTE NOW
For Client: Collect Sample, Test & Receive Test Results

Step 1
Client orders collection Kits by contacting DiaCarta

Step 2
DiaCarta Ships out Collection Kits (NP Swabs, OP Swabs, and Saliva kits)

Step 3
Client collects Patient specimens, enters patient information in DiaCarta LIMS portal (Customer given access to DiaCarta LIMS)

Step 4
Client through LIMS login is enabled to see progress of sample testing

Step 5
Client can receive test results on completion by logging in
Test Process within DiaCarta – Quality Results within 48 hours

Step 1
Specimens delivered to DiaCarta CLIA Lab

Step 2
Specimens Accessioned at DiaCarta Lab via LIMS System (ensuring positive sample identification)

Step 3
Viral RNA Extracted from Collected Specimens

Step 4
RT-qPCR Test completed with extracted viral RNA

Step 5
Results Exported to LIMS System (data reviewed by technologist and approved Medical Director)

Step 6
Results Available to Customers via LIMS System
COVID-19 Total Solution
DiaCarta offers a COVID-19 total solution to support the fight against COVID-19, including the RT-PCR test, antibody IgG test, CLIA lab service, sample collection kits, RNA control and more.

QuantiVirus™ SARS-CoV-2 Test Kit (Detects 3 genes)

QuantiVirus™ SARS-CoV-2 Multiplex Test Kit (Detects 1 gene)
QuantiVirus™ Anti-SARS-CoV-2 IgG Test Kit
CLIA Lab COVID-19 Diagnostics Service

Nasal Sample Collection Kit for COVID-19