SARS-CoV-2 Antigen Rapid Home Test Kit
COVID-19 At Home Test with Simple Nasal Swab.
Research Use Product

Reliable Self-Test

CE/IVD Marked

Easy to Use Nasal Swab

Results in 15 Minutes
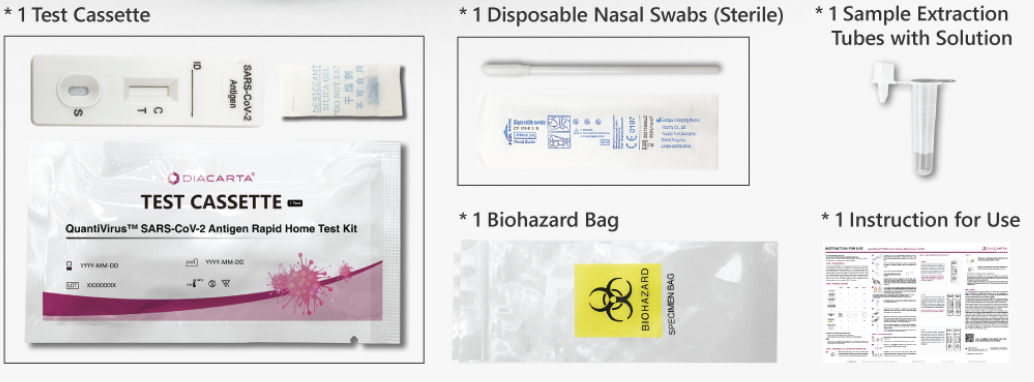
*This image only shows the 1-Test pack size.
Introducing the QuantiVirus™ SARS-CoV-2 Antigen Rapid Home Test Kit
The QuantiVirus™ SARS-CoV-2 Antigen Rapid Home Test Kit is a lateral flow chromatographic immunoassay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2. It is CE/IVD marked.
This test is intended for non-prescription home use with self-collected (unobserved) anterior nasal swab specimens directly from individuals aged 14 years and older, or with adult-collected anterior nasal specimens directly from individuals aged 2 years or older, who are suspected of COVID-19 by their healthcare provider within the first 7 days of symptom onset, or without symptoms or other epidemiological reasons to suspect COVID-19 infection.
Pack size includes 1-test, 2-test, 5-test, and 20-test.
Not for sales in the United States.
COVID-19 Total Solution
DiaCarta offers a COVID-19 total solution to support the fight against COVID-19, including the RT-PCR test kit, antibody IgG test kit, and CLIA lab service.

QuantiVirus™ SARS-CoV-2 Test Kit (RT-PCR Test - Detects 3 genes)

